Multi Radiance Medical MR4 Laser/Stim Package
- Description
Description
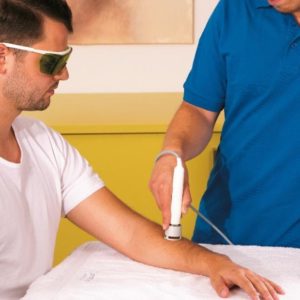
Laser and E-Stim working together like never before
LaserStim™ is the first hybrid emitter that provides both Laser treatment and Electrical stimulation
- World’s 1st FDA cleared laser and e-stim device
- Enables clinician to identify areas of optimal treatment areas
- Provides highly targeted treatments
- 25,000 mW of Super Pulsed Laser Power
- Compatible with acupuncture,muscle trigger probes (optional)
- Compatible with MR4 control unit
- Industry best 2 year warranty
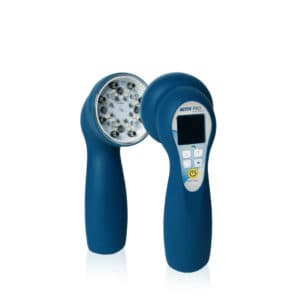
The LaserStim™

Multi Radiance Medical’s LaserStim™ is the first and only FDA cleared device to combine neuro adaptive electrical stimulation and laser in a single emitter, quickly locating the affected areas and then automatically delivering the appropriate dose of energy. The LaserStim is effective and safe to use. LaserStim’s TARGET™ technology provides an interactive neural stimulation by constantly measuring electro-conductivity of treated tissue.
Advantages of MR4 and LaserStim™ with TARGET™
- Wider list of applications than traditional modalities
- Synergistic effect of Multi Radiance therapy provides benefits of light and neuro stimulations
- Faster temporary pain relief
- Assists a clinician in identifying the optimal treatment locations. This is done through our TARGET technology.
- Constantly changing impulses, preventing accommodation of tissue to neuro stimulation
- Deep tissue penetration of photonic energy due to 25W Super Pulsed Laser
- Preset frequencies allow clinician to control depth of therapeutic energy from the surface up to 13 cm (5 inches)
- Insurance reimbursement:simultaneous usage of light and neuro stimulation therapies allows for potential insurance reimbursement
- Patient can feel the treatment
“LaserStim emitter is a positive improvement over the already excellent results I have had with my patients through the years with the Multi Radiance Laser. TARGET feature enables me to identify injured areas quickly and informs me as to what dosages are appropriate. I would highly recommend the MR4 with LaserStim to other practitioners interested in improving outcomes for their patients”.
Dr. David Zymler
Cleveland, OH
* Federal law restricts this device to sale by or on the order of a licensed healthcare practitioner. The purchase of a LaserStim or MR4 LaserStim devices from a third-party seller, such as a public auction website or private, unauthorized individual, should be approached with caution.LaserStim is a prescription device whose use is regulated by U.S. Food and Drug Administration and state and local regulatory agencies. Sellers are required to sell such devices only to individuals qualified to purchase prescription devices (eg, health care professionals). While sellers are also required to confirm the condition of such devices, there is no way to verify the condition of devices purchased from third-party sellers,therefore the warranty for devices purchased from unauthorized LaserStim and MR4 LaserStim distributors cannot be honored and will be void.